Ozempic, Wegovy, Zepbound, and Mounjaro are having a significant impact on human health and wellbeing. These ‘GLP-1 receptor agonists’ reduce appetite and blood glucose levels, and can lead to significant weight loss, in addition to their original use as treatments for Type 2 diabetes.
What is GLP-1?
The story of GLP-1, the hormone having its moment, is tied to the events that took place at the University of Toronto in January 1922. A research group that comprised Fred Banting, Charlie Best, and James Collip, overseen by Professor John Macleod, isolated a factor from animal pancreases that they proposed, based on earlier animal studies, would reduce the hyperglycemia (high blood sugar) of diabetes. On January 11, that material was injected into a 14-year-old boy named Leonard Thompson, who was dying from the toxic effects of a massive blood glucose overload due to his diabetes. He responded positively, but had an immune reaction to some impurity in the injected material. On January 22, a more highly purified preparation was injected. His blood sugar fell into the normal range, and he had little side effect. Insulin was launched into the world.
The discovery of insulin was followed, a year later, by the discovery of a second pancreatic hormone, glucagon, which has the opposite effect to insulin’s: instead of leading to the cellular uptake, storage, and utilization of glucose, it caused the release of glucose from the liver when blood glucose levels fell.
Together, insulin and glucagon are the yin and yang of blood glucose homeostasis. (The term homeostasis was coined by the American physiologist Walter B. Cannon to reflect the body’s ability to maintain constant internal conditions despite varying external ones.) Insulin lowers blood glucose, and glucagon causes its replenishment. They are released from the pancreas in response to hyper- or hypo-glycemia, respectively.
Another factor, which had been predicted, came to light in the 1980s. Like insulin and glucagon, it is a peptide hormone. However, it is produced in the gut after a meal. This factor has several effects. It causes the pancreas to produce a higher level of insulin in the presence of a high blood glucose levels, such as happens after a meal. And it suppresses the production of glucagon by the pancreas. It is part of blood sugar homeostasis. This hormone looks very similar to glucagon. In fact, it is a product of the same gene, although it has the opposite effect. This is the ‘Glucagon-Like Peptide’, GLP. There is more than one Glucagon-Like Peptide; the one that is part of this story is GLP-1.
The dramatis personae of blood glucose homeostasis
It’s important to keep blood glucose levels within a normal range. Too high glucose, hyperglycemia, over an extended time leads to damaged blood vessels, tissues, and organs, and to the dangerous metabolic condition ketoacidosis. Too low glucose and we pass out (the brain depends almost exclusively on glucose for energy).
When blood glucose is rising, as after a meal, insulin is released from the beta cells of the pancreas. Insulin has several target tissues, including muscle, liver, and adipose (fat) tissue. It’s overall effect is to cause those tissues to take up glucose from the blood and either use it as an energy source, or convert it into glycogen (liver and muscle) or fat. It is sometimes referred to as ‘the storage hormone’.
But hours after our last meal, blood glucose levels begin to fall, and this stimulates the release of glucagon from the alpha cells of the pancreas. Of the three actors, glucagon, insulin, and GLP-1, glucagon appears to have the simplest role to play. It is primarily a ‘hypoglycemia factor’ — it is secreted from the pancreas when blood glucose levels fall below the normal range, and it stimulates the liver (and to a lesser extent the kidneys) to release glucose. The source of that glucose is both the ‘starch-like’ storage polymer glycogen (which has been brought up in several other posts), and, under starvation conditions, degraded proteins that are converted to glucose in the liver.
When our guts are full after a meal, cells there produce GLP-1, which has a range of target tissues. It slows the emptying of the gut after a meal, creating a feeling of ‘fullness’. In the presence of high blood glucose, it triggers the pancreas to produce a higher level of insulin and less glucagon. It transmits a signal to cells of the hypothalamic nucleus of the brain to make us feel satiated. It sounds like a cure for overeating, but it is short-lived in the blood, so administering it intravenously is not an effective treatment for the overweight. But the central idea is a good one.
GLP-1 receptor agonists arose from the effort to help Type 2 diabetics. Type 2 diabetics produce insulin, but their target cells have lost their sensitivity to it. It would be useful for Type 2 diabetics to increase the insulin response by the pancreas, and from its known ability to do this, GLP-1, or its receptor agonists, were tried. Indeed, they did help control blood glucose in Type 2 diabetics. What was observed, was that such people also lost weight. This led the way to the testing of these drugs to stimulate weight loss.
The genetic origins of glucagon and GLP-1
Glucagon and GLP-1 are peptides, which are usually defined as being proteins containing fewer than 40 amino acids in a chain. Like other proteins, they are genetically coded in genomic DNA. As it happens, by the same gene. That gene is transcribed (copied into a messenger RNA) and translated (the mRNA dictating the sequence of a protein). The resulting protein is 180 amino acids long, and contains both glucagon and the glucagon-like peptide GLP-1. The awkward name of that protein, ‘preproglucagon’, reflects the fact that it is heavily edited into the final products (glucagon and GLP-1, and in some tissues, other peptides also).
One of the tissues in which preproglucagon is produced are the alpha cells of the pancreas. There, it is edited into glucagon by removal of most of the preproglucagon protein chain. Glucagon contains only 29 amino acids of the original 180. Preproglucagon is also produced in certain cells of the gut, where a different type of editing produces GLP-1, which has 30 amino acids.
Glucagon and GLP-1 come from different parts of the preproglucagon protein, but their amino acid sequences are very similar. This is probably because the preproglucagon gene was created by the duplication of a shorter genetic sequence during evolution, and then mutated. In the one-letter code used for the amino acids, the sequences of the two peptides are:
Glucagon: HSQGTFTSDYSKYLDSRRAQDFVQWLMNT
GLP-1: HAEGTFTSDVSSYLDSRRAQDFVQWLMNT
There are only four amino acid differences.
What’s in a Name?
And so we come to the GLP-1 receptor agonists, Wegovy, Ozempic, Zepbound, and Mounjaro (and others). Like many hormones, they interact with cells by binding tightly to specific cellular receptors. They bind to the same receptors as GLP-1. (The latter two also bind another receptor involved in glucose homeostasis.) They activate the cells they bind the same way that GLP-1 does. The name for a factor that does this is ‘receptor agonist’. (As compared to ‘receptor antagonist’, which blocks binding of the normal agent to its receptor.) Despite their structurtal similarity, glucagon and GLP-1 bind to different cellular receptors, and have different cellular effects.
Like GLP-1 itself, the receptor agonists resemble glucagon’s structure for the important part of their amino acids sequence. In the case of Ozempic and Wegovy, they also have a long fatty acid chain added, and this give them an important property: they bind to human serum albumin, which is a long-lived protein in the blood, and because of this, they also have a long half-life, in contrast to GLP-1 itself.
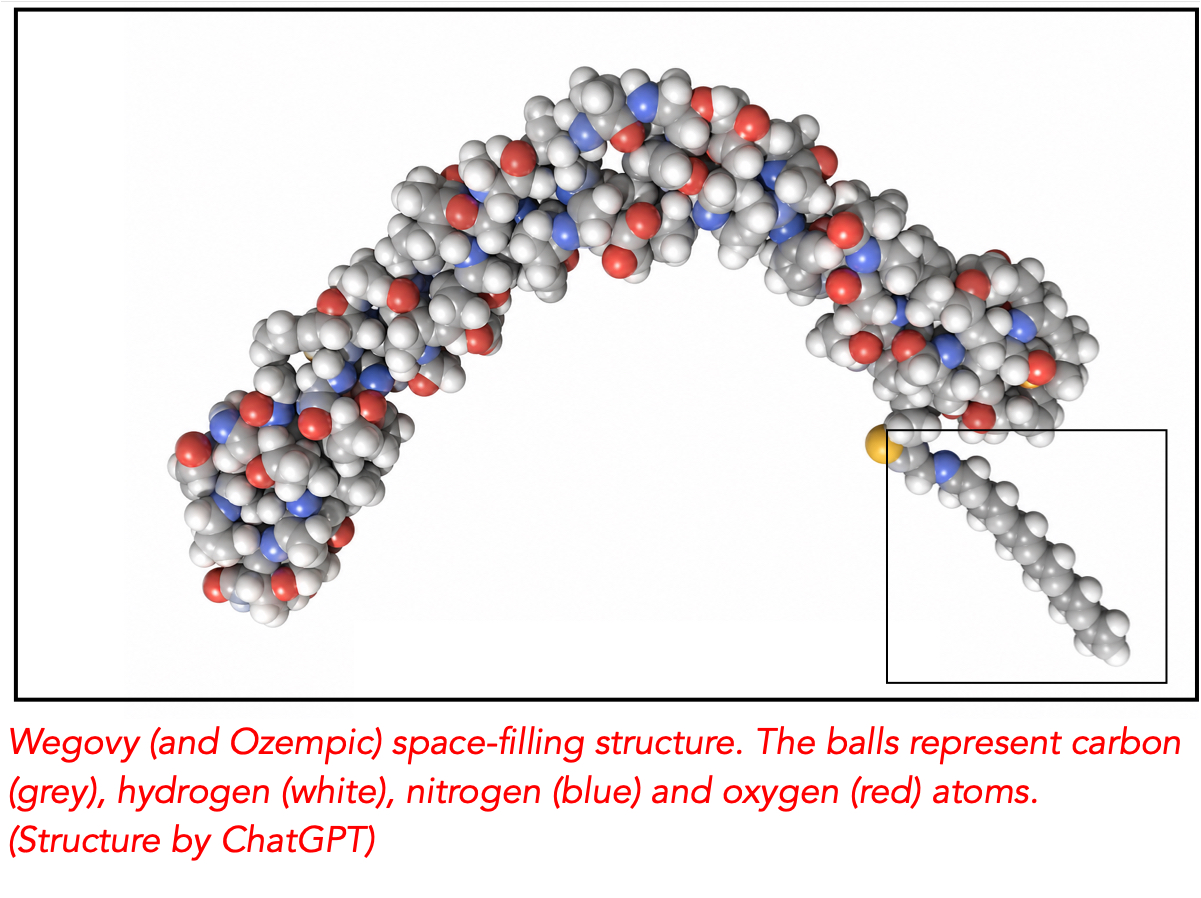
The Wegovy (and Ozempic – they are the same) space-filling structure is shown at the top of the post. The structure resembles GLP-1 in the important left-hand (amino terminal) end. The most important difference with GLP-1 is the attachment of an aliphatic (fat-loving) chain (grey and white atoms) to the amino acid chain (box in lower right-hand corner).
The skeleton structure below illustrates this. It shows how the GLP-1 receptor agonist can have both of its ends in contact with the cellular surface, one with the GLP-1 receptor, the other with another cellular component. The lipophilic side chain (the box) has an affinity for part of human serum albumin, which protects it against degradation in the blood.
The future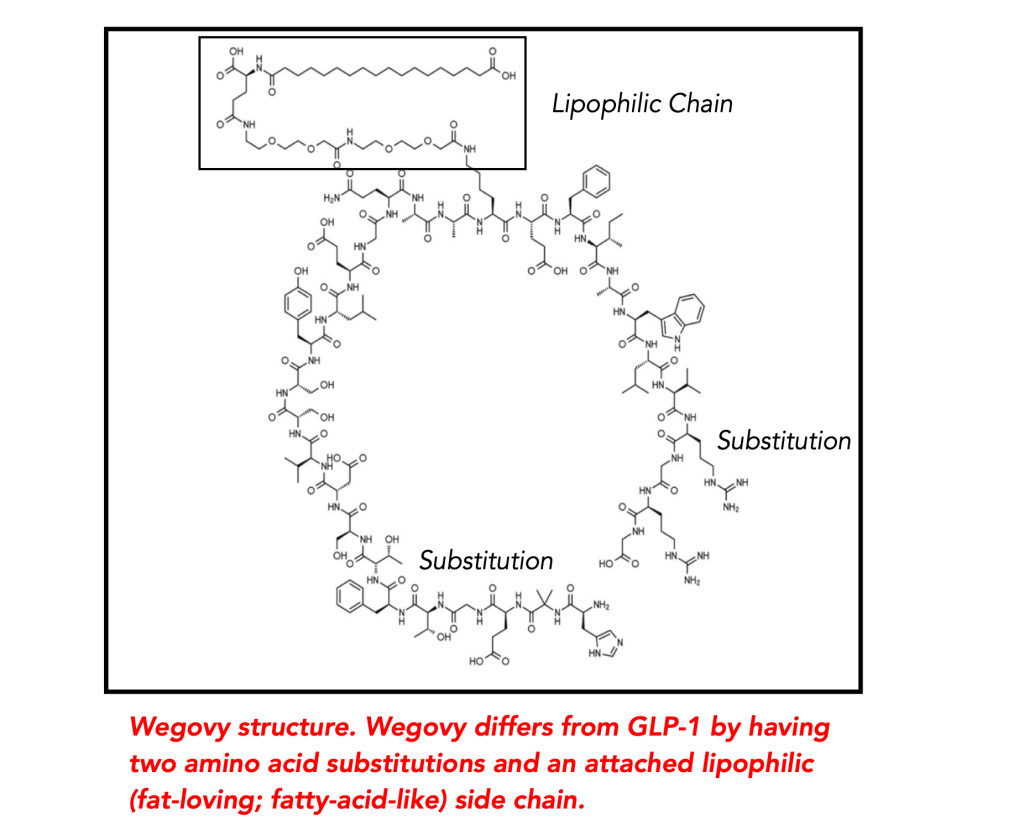
The long-term effects of the GLP-1 receptor agonists are still not well understood, and it appears that most people have to take them continuously to keep the weight off. Nevertheless, this class of pharmaceuticals has been received joyously by people who have previously struggled unsuccessfully to lose weight.